FULL PAPER (日本語ー要旨のみ)
Information Processing Effect on
Neuropsychiatry, Neuropsychology & Behavorial Neurology, (Journal Name Changed to: Cognitive and Behavioral Neurology) 3:2, 80-97; 1990.
(1) Department of Psychiatry, New York Medical College, Valhalla, New York, U.S.A.
(2) Department of Psychiatry, Tokai University School of Medicine, Isehara, Japan
(3) Department of Psychiatry, Minakawa Memorial Hospital, Samukawa, Japan
This research was supported by a grant from the Matsumae International
Foundation, Tokyo, Japan, and was Dr. Berger's Ph.D. dissertation at Tokyo University. ABSTRACT
Information processing was tested by comparing saccadic reaction times of 13 schizophrenics and 13 normal controls under conditions where there was information provided about the duration of a warning signal to those where there was not. Saccadic reaction time measured by electro-oculogram was studied in order to minimize complicating variables associated with prior studies using manual reaction time.
Supporting prior research, this study found that schizophrenics do worse when there is prior information about warning signal duration than when there is not, while in controls this was reversed. Significant enhancement of this effect with increasing age and a possible normalizing effect with greater neuroleptic dose were also found. The major limitations of this study, however, included a small N and uncontrolled medication.
The authors conclude that (a) there is a schizophrenic deficit in the processing of information in order to establish a mental set necessary for preparation, (b) this deficit may be useful in confirming the diagnosis of schizophrenia in older age groups, and (c) effects of antipsychotic medication on information processing require further study. They hypothesize that the effects of neurotransmitters on information processing and time perception, abnormalities in the frontal lobes, and information processing steps measured by evoked potential may play a role in impaired information processing in schizophrenia.
INTRODUCTION
(Note: Only selected figures are available on-line.)
Reaction time (RT) studies, reviewed by Nuechterlein (1977), have been consistently replicated in schizophrenia (SZ) research. A significant group of studies
in this regard include those that investigated the ability to prepare for RT by providing prior time information about foreperiod duration. Forperiod is the time between onset of the warning signal and the imperative stimulus. Huston et al.(1937) reported on simple auditory-manual RT that significantly differentiated patients with chronic SZ
from normals. SZ patients had a longer mean RT, and whereas in normals the
RT was faster when the duration of preparatory intervals was held constant, in
SZ patients this difference did not persist. This indicates that SZ were unable to
profit from this regularization. Presumably, the regular series allows the subject
to learn when to expect the imperative stimulus and to prepare maximally to
react to it by establishing a "mental set."
Rodnick and Shakow (1940) replicated these results in a simple visual-manual
RT study. Although Huston and Senf (1952) found the greatest impairment
in set in chronic patients, Iesser degrees of impairment could be found in patients
with early SZ or depressive psychoses and to a smaller extent in neuroses. This
last study, along with one done by Zahn and Rosenthal (1965), who found considerable overlap of acute SZ and non-SZ cases, have questioned the diagnostic
utility of this measure in acute cases. In chronic cases, however, there is a statistically significant degree of discriminability using this test. Tizard and Venables (1956) found excellent discrimination in impairment of set between chronic SZ and mentally retarded adults, suggesting that this impairment in set reflects something other than an intelligence factor.
In a visual-manual RT experiment with a variable auditory foreperiod where
pitch of the sound acted as a prior time information about foreperiod duration,
Mo and Kersey (1980) compared chronic SZ patients to alcoholics. When pitch
was correlated with foreperiod duration, so that there was prior time information
about foreperiod duration, SZ patients performed worse than when there was no
correlation although it did not reach the 0.05 Ievel of significance. Alcoholics, on
the other hand, did not differ in this regard. They concluded that in SZ patients
there was a suppression of preparation by expectancy, and they hypothesized that
the motor preparation itself decreased the ability of their data to reach
significance.
The purpose of the present study is to clarify the effect of prior time
information on RT and to relate the results to current neuropathophysiologic studies in
SZ patients. In order to do this, we propose the study of foreperiod in the RT of
saccadic eye movement. The advantages of this are many.
(a) There has been a consistent finding of a slowed simple manual RT in SZ
patients (Nuechterlein, 1977). Saccadic latency, in contrast, has been shown in at
least three studies to be normal in SZ patients, whether or not there was impaired
smooth pursuit (Iacono and Tuason, 1981; Levin et al.,1981,1982). If saccadic
RT is affected by foreperiod conditions, then it is not likely due to a function
inherent in the saccadic system itself.
(b) In the above-cited studies, visual or auditory stimuli are coupled to manual
responses, thus utilizing a more complicated system of sensory motor integration
than the visual-oculomotor saccadic response. If there is a basic SZ deficit in the
handling of sensory data, then one would want to keep the processing of this data
to a minimum when testing for such a deficit. The superior colliculus can directly
relate visual input to oculomotor commands and the saccadic system is a discrete
control system whose velocity is not under voluntary control. Eye muscles are
faster than those used in a manual response, do not pull against gravity, and
oculomotor neurons fire much faster than spinal motor neurons (Kandel and
Schwartz, 1981).
(c) Unlike simple RT tasks, in the saccadic task the imperative stimulus is
determined by a change in location (the target moves) rather than by the
introduction of a stimulus.
(d) Saccades are considerably less dependent on proprioception than manual
responses and proprioceptive deficits have long been suspected in SZ (Iacono and
Tuason, 1981).
(e) Saccades recorded via electro-oculogram will provide highly accurate
information for the measurement of RT (Iacono and Tuason, 1981; Mialet and
Pichot, 1981; Lipton, 1983).
(f) Comparison of foreperiod effect on the saccadic system to that of the
manual response system will provide evidence to determine whether this effect is
unique to a particular system or common to both.
Antipsychotic drugs have not been found to affect simple manual RT, foreperiod effect on manual RT, saccadic latencies, or smooth pursuit eye movements
(which include an initial saccade and sustained attention to a target) (Held et al.,
1970; Neuchterlein, 1977; Lipton, 1983; Holzman, 1987). One study concluded
that RT got better on phenothiazines although this was not tested for statistical
significance (Held et al.,1970). The present study will further address this issue
of drug effect on RT.
Study of the foreperiod information effect on RT may tap multiple steps in
the processing of information. The prefrontal cortex governs planning, abstract
thinking, the ability to change expectations in response to new data, and
sequential thinking among others (Foster,1980; Andreasen,1988). Prior time
information about foreperiod may test frontal lobe processing of this information since subjects will be required to create new expectations about foreperiod duration in response to new data and then to react.
In a review of SZ, Andreasen (1988) delineates various tests of brain structure
and function that have been found to be abnormal in SZ. Computed tomography
(CT) and magnetic resonance (MRI) brain imaging studies have shown increased
ventricular size that does not change with age, stage of illness, or drug treatment.
The most consistent correlate with CT abnormalities is cognitive impairment.
MRI reveals decreased frontal size, position emission tomography (PET) scan
shows decreased prefrontal metabolism, and regional cerebral blood flow (rCBF)
studies find decreased activity of the prefrontal lobes when SZ patients do the
Wisconsin Card Sorting Task or the continuous Performance Test, both of
which require the ability to change expectations to new data. SZ patients seem to
have difficulty activating the prefrontal lobe when they need it. Cytoarchitectural
studies of the frontal system have found changes that are compatible with an
early developmental abnormality, suggesting a pathologic process that begins
before the onset of clinical illness. The information processing task in the present
study will likely tap the functioning of the prefrontal cortex.
Another area that tests information processing is the evoked potential studies,
recently reviewed by Holzman (1987). The P300 wave, a later occurring wave on
EEG during evoked potentials, reflects the cognitive work done on the stimulus,
and the N 100 wave, which is an early wave reflecting stimulus registration, have
been found to be abnormal in SZ patients. The P300 wave is attenuated,
suggesting difficulty extracting information from the stimulus. The NIOO wave has
shown less variation and heightened amplitude, suggesting impaired subcortical
filters with abnormal amounts of information reaching the cortex. The
information processing task in the present study will likely be handled by those steps
measured by evoked potentials.
METHODS
Subjects
Patients
Thirteen patients who gave informed consent were studied from a long-term
inpatient psychiatric hospital. All patients were ethnically Japanese with identical
cultural backgrounds. A11 patients met DSM-IIIR criteria for chronic
schizophrenia and none had concomitant diagnosis of mental retardation. Five patients
were of the paranoid subtype, three were disorganized, two were residual, two
were in acute exacerbation, and one was undifferentiated. Patients were
excluded from the study if they had electroconvulsive therapy (ECT) during the last
10 years or were currently on a benzodiazepine or other sedative-hypnotic, or if
they were unable to cooperate or understand the instructions. The patient group
consisted of nine males and four females. The average age was 41.4 years-old with an
age range of 32 to 48 years.
All patients were on a high-potency neuroleptic and 9 of I 3 were
concomitantly on an anticholinergic. Details of medication, symptomatology, hospital
duration, and Brief Psychiatric Rating Scale (BPRS) ratings are provided below.
All clinical determinations were done by the physicians currently caring for the
patients without a knowledge of RT scores.
Controls
Thirteen controls volunteered from the staff at the medical center and were
age- and sex ratio-matched to the patient group. Average ages for controls and
patients were not significantly different. Nine controls were male and four were
female. The average age was 43.4 years with an age range of 35 to 50 years.
Exclusion criteria were the same as in the patient group. In addition, any control with
a history of a major psychiatric disorder or a first-degree relative with such a
history was excluded.
Detailed questionnaires were given to all subjects in order to rule out a history
of neurologic, vestibular, visual, auditory, or medical condition that might
interfere with the study. Glasses were required to be worn if needed. Neurologic
screening examinations were also done on all subjects with particular attention
to hearing, vision, and extraocular movements to insure that these systems were
intact.
Procedures
Subjects were seated and were instructed to watch a horizontal display panel
0.5m away and at eye level. Heads were stabilized at the chin and forehead. The
target was a 1.0 cm diameter circular-red light constantly visible at center field
that moved abruptly in the horizontal plane to the right or left in 20' of visual
arc. Stimulus proceeded instantaneously without delay at the end of an auditory
foreperiod provided by a speaker situated immediately behind the stimulus
display.
The sound frequency varied in three measures (250, 500, and 1,000 Hz) as well
as its duration (1,3,and 5 s). In the correlated (COR) trials, frequency and
duration covaried so that frequency could function as prior time information about
foreperiod duration. The covarying combination that was used was 1s with 250
Hz, 3s with 500 Hz, and 5s with 1,000 Hz. In the uncorrelated (UNC) trials,
frequency and duration varied randomly so that subjects could not pre-estimate
the foreperiod duration.
In both COR and UNC conditions, the same number of each of the three
frequencies and durations and in the same order within each condition set were
presented to all subjects. Thirty of each COR and UNC trials were given. Both
COR and UNC conditions were divided into 15 trails to the right and 15 to the
left. Approximately 5-10s separated each trial, and 1-2 min rest was provided
between direction changes and 5 min between the COR and UNC trial sets.
In order to study the effects of fatigue and practice on RT, sequences of the
COR and UNC trial sets were reversed in one-half of both the patient and control
groups. The stimulus direction reversed within these sets so that in each of the
control and patient groups, there were four subgroups with differing trial set
sequences.
All subjects were provided with instructions before each set of trials as to
stimulus direction and the nature of the frequency-duration correlation. Three
unrecorded practice trials were provided before each direction change. Subjects were
also instructed to refrain from blinking, to relax their facial muscles, to watch
only the target, and to respond as quickly as possible to follow the target when it
moves. Visual distraction was minimized by turning out the lights. The sound
level was audible in the adjoining room, where equipment was operated, and all
subjects reported that the sound was readily audible.
Eye movement was recorded via electro-oculogram (EOG) with standard EEG
electrodes placed, two at the inferior lateral aspects of each eye, two at the superior
medical aspects, and a midforehead ground. A Nihon Kohden standard EEG
machine was used with settings adjusted as necessary to maximize the EOG and
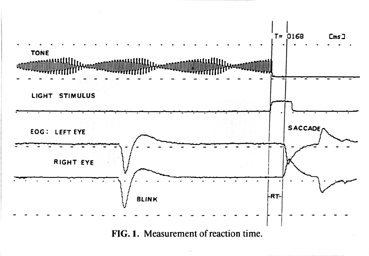
to minimize EEG and EMG noise. Saccades were clearly distinguished in all cases
from the EEG and EMG (See Fig. 1 below). Electrode position readily enabled distinction of blinks and vertical saccades from horizontal saccades. Horizontal saccades were always registered in opposing directions on the EOG, while the others were always in the same relative direction. Excessive blinking during the foreperiod or stimulus
could be monitored and instructions given to the subject to refrain.
Four channels were used for recording. Channel 1 recorded foreperiod tone,
channel 2 recorded the stimulus displacement, and channels 3 and 4 recorded left
and right eye EOG, respectively. All four channels were simultaneously
recorded on a data recorder for future RT measurement.
Data Analysis
RT measurement was performed via display of recorded data on a polygraph.
Vertical bars were manually adjusted to separate the interval from stimulus
initiation to saccadic reaction, while a computer determined the RT in milliseconds.
This procedure enable accurate RT measurement to within 2ms. Blinks
occurring near the end of the foreperiod resulted in exclusion of that trial in order to
insure that a blink did not interfere with stimulus recognition.